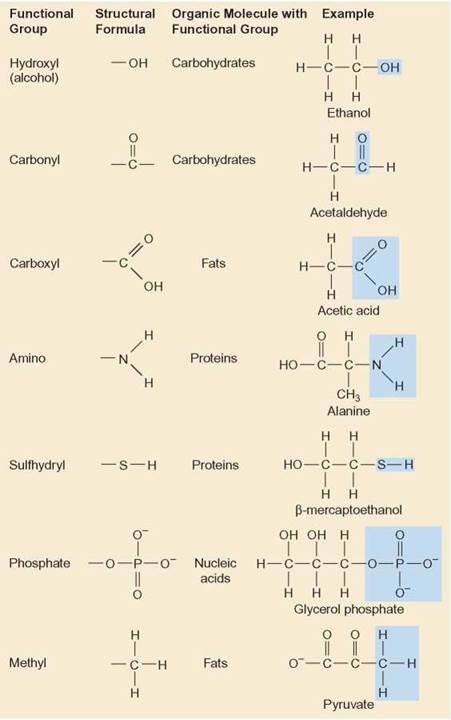
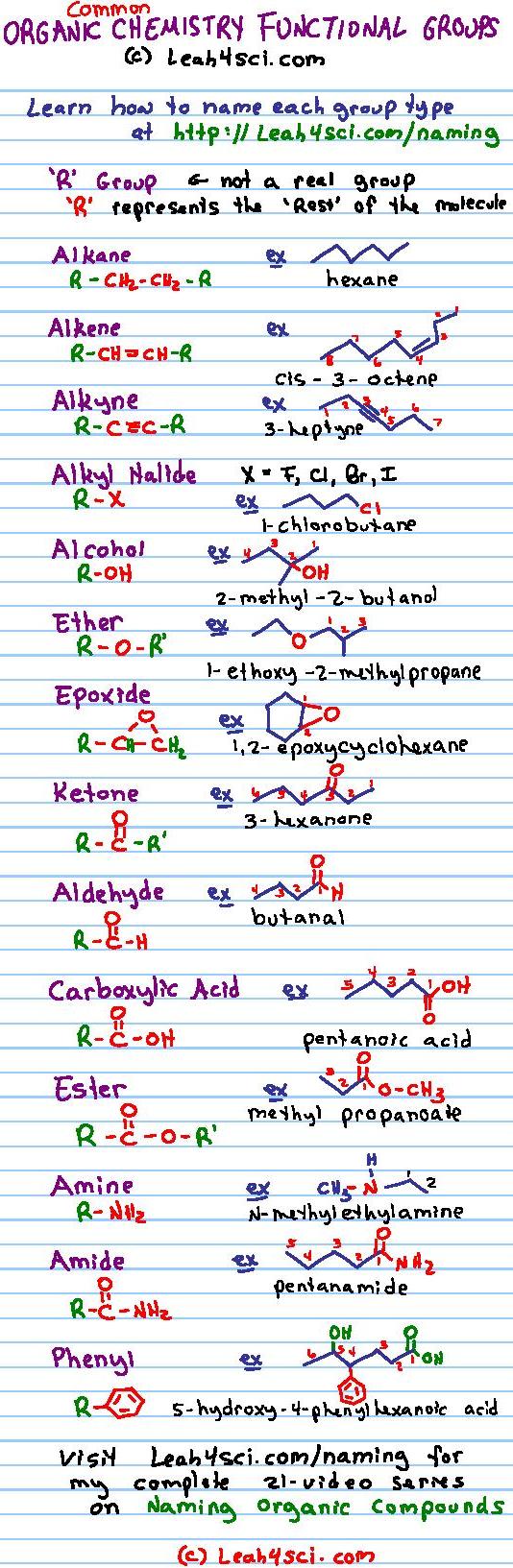
This actually is the relatively easy part of organic chemistry which then goes on to consider the reactions used in the synthesis of organic compounds such as the adding of functional groups to organic molecules. In addition are various hydrophilic linkages such as ethers (i.e., C- O- C), ester linkages (as found holding together fats, i.e., triglycerides), phosphodiester linkages ( nucleic acids), glycosidic linkages ( disaccharides and polysaccharides), and peptide bonds ( polypeptides/ proteins).Įarly on in studying organic chemistry one learns the various functional groups along with naming conventions. We will go through several examples for more details about the naming rules.Hydrophilic functional groups include hydroxyl groups (resulting in alcohols though also found in sugars, etc.), carbonyl groups (giving rise to aldehydes and ketones), carboxyl groups (resulting in carboxylic acids), amino groups (i.e., as found in amino acids), sulfhydryl groups (giving rise to thiols, i.e., as found in the amino acid cysteine), phosphate groups (as found in nucleic acids and phospholipids), etc. Table 2.3 Naming Priorities of Common Functional Groups The groups in the subordinate table have no difference in terms of priority, and they are usually listed in alphabetic order. The order of the groups listed in Table 2.3is based on the decreasing order of priority, where the carboxylic acid group is in the highest priority. The “suffix” is used to indicate the name of the parent structure, and the “prefix” is for the substituent. If the compound includes more than one functional group, the one with the highestpriority is the “parent structure” and determines the “parent name” the other groups will be regarded as “substituents”. functional groups are chemical motifs, or patterns of atoms, that display consistent function (properties and reactivity) regardless of the exact molecule they are found in. A protective group (also referred to as protecting group) is a reversably formed derivative of an existing functional group in a molecule. Assign stereochemistry, E/Z or R/S, as necessary (details in Chapter 5).įor naming purposes, the functional groups are assigned with priorities (Table 2.3).Some organic functional groups are as follows: Carboxylic acid: COOH. The other groups are named as substituents by using the appropriate prefixes. Functional group: The atom or the group of atoms by which the characteristic reactions of organic compounds are determined, that atom or group of atoms is called the functional group.Number the chain from the end closest to the highest functional group. The concept of functional groups forms a basis of organic chemistry, medicinal chemistry, toxicity assessment, spectroscopy and also chemical nomenclature. 
Some of the functional groups taught in school chemistry courses include halogens. For example, one such functional group is the hydroxyl group, consisting of an oxygen atom and hydrogen atom joined to each other. The foundational functional group is the alkane.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
